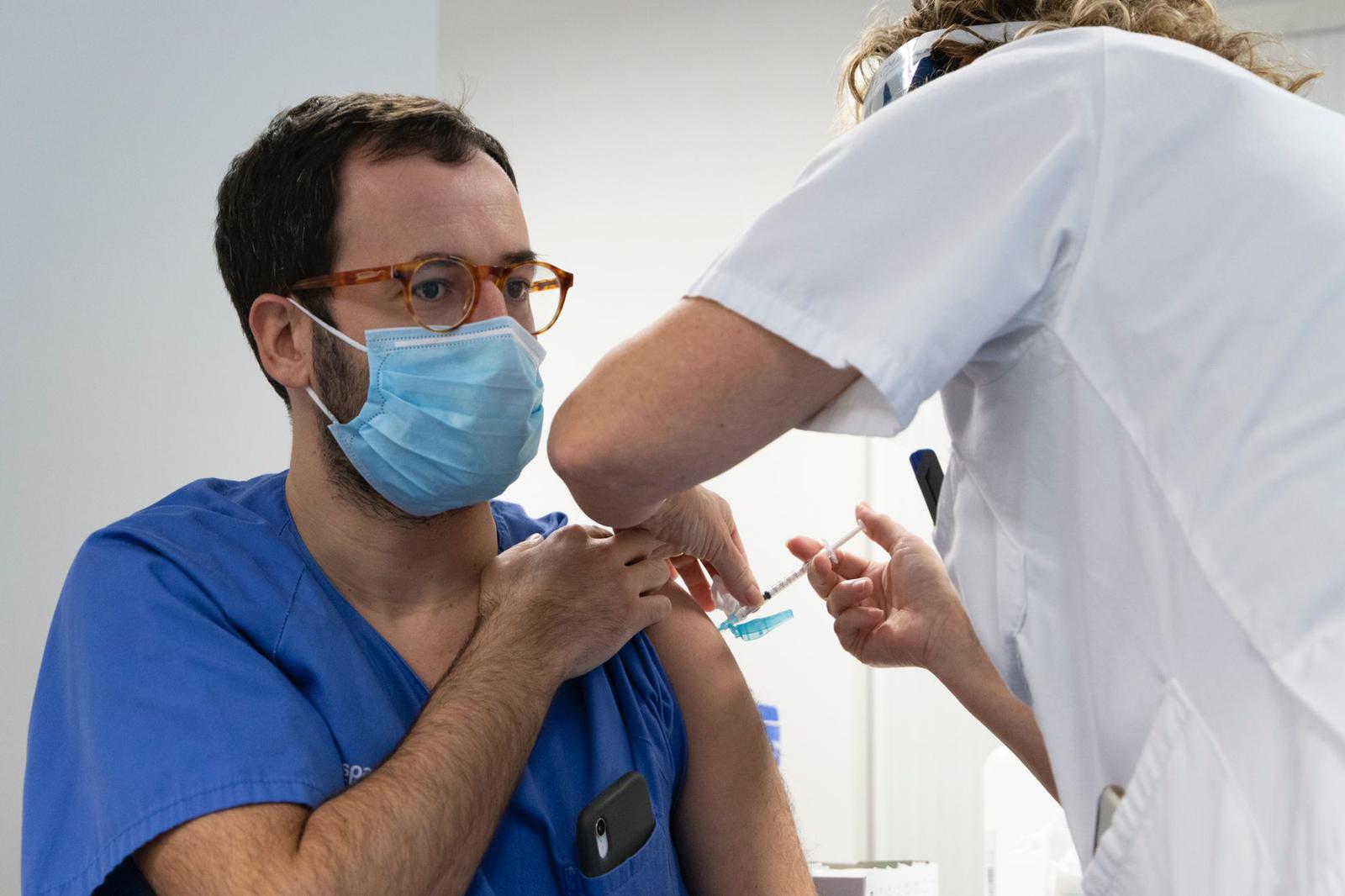
THE Ministry of Health yesterday delayed opening up the AstraZeneca vaccine to the 55-65 age group as they evaluate reported adverse side effects.
In yesterday’s meeting, the committee refused to leave with a definitive answer to whether they would begin to administer the vaccine to this age group as they need to investigate claims that it causes a number of severe health issues.
“We will wait until there is an evaluation and opinion of the European Medicines Agency (EMA) regarding the adverse effects of this vaccine,” said a statement fro the Ministry of Health.
Currently the age limit outlined in the Oxford vaccine’s program is 55, with those older issued with other vaccine options such as the Moderna and the Pfizer brands.
Representatives from Madrid, Catalonia and Andalucia have pushed the Ministry to make the AstraZeneca vaccine available to the more vulnerable age group.
A discussion was due to be had in April regarding the age limit but the Minister of Health, Carolina Darias, decided to push the meeting forward, however the results remain inconclusive.
As part of the British vaccines distribution plan, non front line health workers, essential workers and teachers were first to be given the injection as long as they were under the age of 55.
Spain’s government has been put under pressure to make a decision on the AstraZeneca vaccine after numerous European countries have banned the vaccine due to possible fatal side effects.
Denmark, Norway and Italy have banned the vaccine after isolated cases of fatal blood clots occurred several days after receiving the jab.
Italy and Austria followed suit after the death of a 49-year-old woman died from complications related to a blood clot 10 days after getting the vaccine.
Experts from the EMA have been cautious to link the vaccine to the deaths, “There is currently no indication that vaccination has caused these conditions, which are not listed as side effects with this vaccine,”
However it has led Spain to re-evaluate its distribution of the AstraZeneca vaccine before further studies are carried out.